Aluminium has been widely used in industry for decades. The industrial interest in cast aluminium alloys is gradually increasing due to its low cost and excellent fluidity, however, their poor tribological properties and low corrosion resistance notably reduce their aplication.
That's when Cidetec enters the scene.
In general, tribological and corrosion properties of aluminium and its alloys can be improved by anodising. This electrochemical technique increases the thickness of the natural oxide layer, converting the surface of the aluminium alloy into a ceramic coating (Al2O3) with improved hardness, wear and corrosion resistance but comercial aluminium cast alloys contain intermetallic particles that negativelly influence the general anodizing response.
Cidetec has studied the sulphuries acid anodizing of EN AC-46500 cart alluminium alloy and results from this work may assist in selecting the most appropriate experimental conditions for the development of high quality anodic oxide layers on this cast alloy that would made possible the implementation of aluminium in areas where it was not considered possible hitherto.
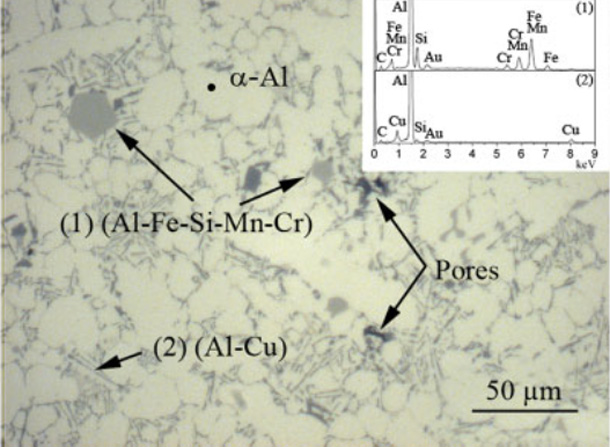
Low magnification image of sample microstructure.

Detail of areas with different morphologies in aluminium alloy matrix.
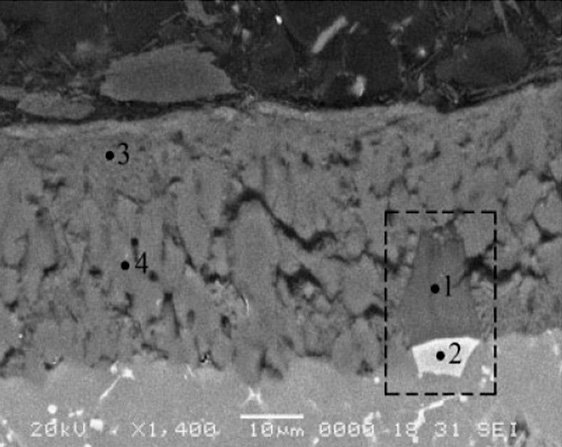
a SEM image of cross-sectional anodic alumina layer formed on EN AC-46500 aluminium alloy after anodising in 15 vol.¯%H2SO4 electrolyte at 0uC and 5,0 A dm22 for 30 min. Highlighted area corresponds to partially anodised Al–Fe–Si–Mn–Cr particle.
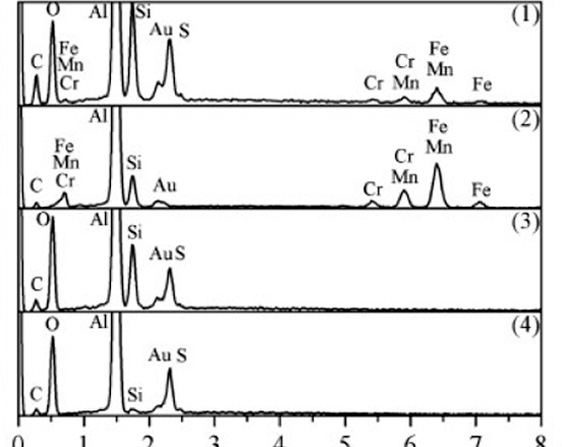
EDS spectrum from different regions marked in a (small Au peak is due to metallic gold that was sputter coated on anodic alumina layer to reduce electrical charging during SEM measurements)
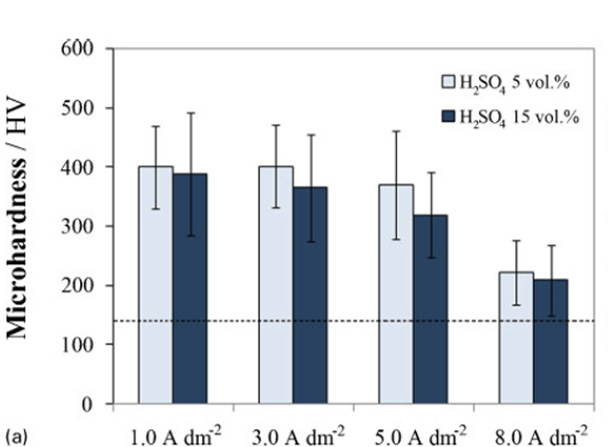
Effect of electrolyte concentration and current density (at 0uC for 30min)
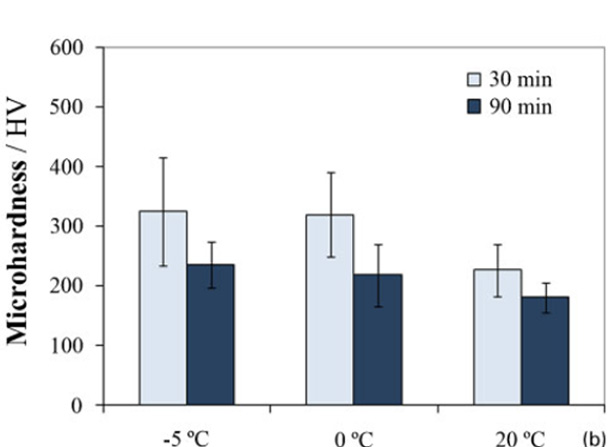
Effect of temperature and anodising time (in 15 vol.¯%H2SO4 electrolyte at 5,0 A dm¯2) Vickers microhardness of anodic alumina layers formed on EN AC-46500 aluminium alloy after anodising under different conditions